Reviewed by Joost Boormans (Erasmus MC Cancer Institute, Rotterdam, the Netherlands)
Data from a recent study support the use of a urinary methylation-based assay, i.e. Bladder EpiCheck®, to accurately identify recurrence in patients with high-grade non-muscle-invasive bladder cancer (NMIBC) treated with intravesical instillations [1].
The study retrospectively evaluated 374 patients with high-grade NMIBC, including 106 patients with carcinoma in situ, under intravesical instillations (BCG or mitomycin C). The patients were followed up for 1 year by voided urinary cytology, followed by white-light cystoscopy and biopsies, where necessary. The referral for cystoscopy and biopsy was based on the cytology results according to The Paris System for Reporting Urinary Cytology (TPS) [2]: patients were referred for cystoscopy if falling in one of the following categories: high-grade urothelial carcinoma (HGUC), suspicious for high-grade urothelial carcinoma (SHGUC), or atypical urological cells (AUC). Bladder EpiCheck was performed together with cytology in all cases. An EpiCheck score ≥60 was considered positive.

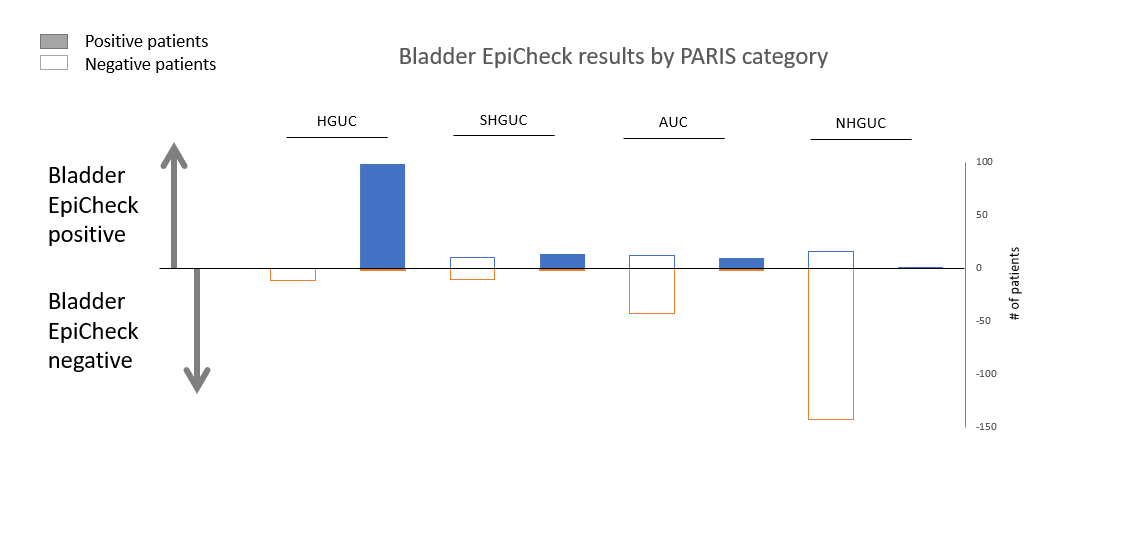
Follow-up according to diagnostic categories for The Paris System for Reporting Urinary Cytology (TPS) (N=374 patients)
Data from this study showed that a positive Bladder EpiCheck results significantly correlated with HGUC (odds ratio = 3.957 vs. SHGUC, p=0.003) and SHGUC (odds ratio = 3.791 vs. AUC, p=0.003). A negative Bladder EpiCheck result significantly correlated with NHGUC (odds ratio = 3.925, p=0.0003). When compared to pathology, Bladder EpiCheck demonstrated sensitivity of 95%, specificity of 85%, NPV of 97% and PPV of 76%. Sensitivity, specificity, negative and positive predictive value of Bladder EpiCheck were higher in HGUC vs SHGUC.
In conclusion, these data support that Bladder EpiCheck has high correlation with urine cytology in the diagnosis of SHGUC and HGUC
Share this on your favourite network:

